 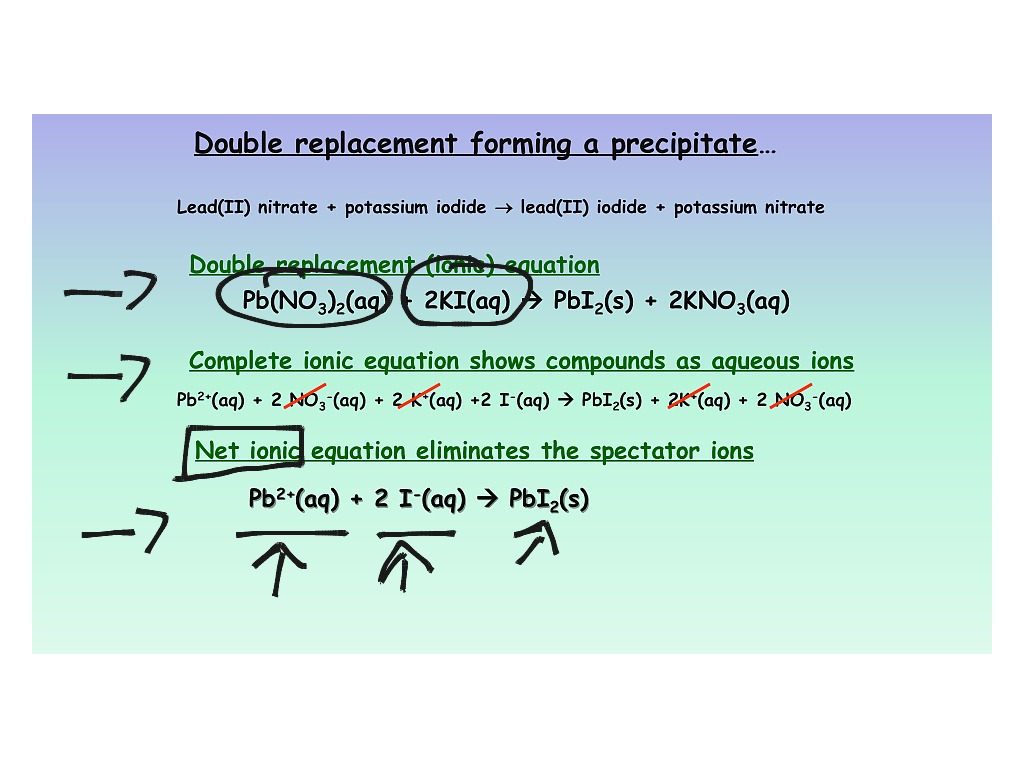
Equations with all the soluble reactants and products dissociated into ions are called. Subscripts are not needed to describe the state of the matter, because all ions are in aqueous. As you know, soluble ionic compounds exist as ions in solution. However, we know that water-soluble ionic compounds dissociate into ions. Ionic equations show species reacting as their ionic components. 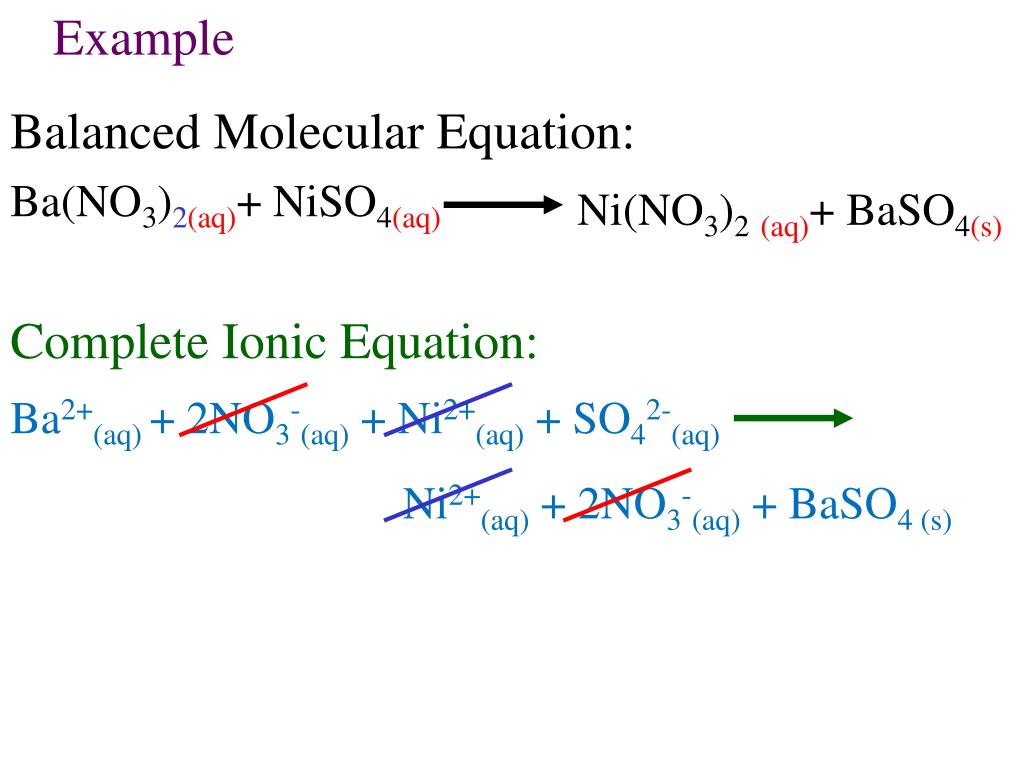
What information can be obtained from a complete ionic. Youre dealing with a double replacement reaction in which two soluble ionic compounds react in aqueous solution to form an Insoluble solid. Then write the ionic equation, showing all aqueous substances as ions. SIDE NOTE You will sometimes see the hydrogen ion being replaced by the hydronium ion, #"H"_3"O"^(+)#. A complete ionic equation consists of the net ionic equation and spectator ions. Write and balance the molecular equation first, making sure that all formulas are correct. Write the balanced formula, complete ionic, and net ionic equations for each of the. To get the net ionic equation, simply remove the spectator ions Write a balanced chemical equation for any reaction that occurs. You can tell that this is the case because sodium chloride, #"NaCl"#, one of the two products of the reaction, is soluble in aqueous solution. Below is the complete ionic equation: (8.4.2) Ba 2 + ( aq) + 2 Cl ( aq) spectator + 2 Na + ( aq) spectator + SO 4 2 ( aq) BaSO 4 ( s) + 2 Na + ( aq) spectator + Cl ( aq) spectator Complete Ionic Equation Equation 8.4.2 is rather cumbersome and includes so many different ions that it may be confusing. The sodium cations and the chloride anions act as spectator ions because they are present on both sides of the chemical equation as ions. Now, when these two solutions are mixed, the hydroxide anions produced by the strong base and the hydrogen ions produced by the strong acid will neutralize each other to produce water. If all species are spectator ions, please indicate that no reaction takes place. #"HCl"_ ((aq)) -> "H"_ ((aq))^(+) + "Cl"_ ((aq))^(-)# Show the complete ionic and net ionic forms of the following equations. This tells you that the two reactants will dissociate completely in aqueous solution to produce cations and anions. Write three equations (complete chemical equation, complete ionic equation, and net ionic equation) that describe this process. If the acid and base are equimolar, the pH of the resulting solution can be determined by considering the equilibrium reaction of A with water. It is just there to balance out the charge because you can't have an ion just by itself - you must always pair an ion with another one of opposite charge so that the overall charge is zero.The thing to recognize here is the fact that you're dealing with a neutralization reaction that features sodium hydroxide, #"NaOH"#, a strong base, and hydrochloric acid, #"HCl"#, a strong acid. When a weak acid and a strong base are mixed, they react according to the following net-ionic equation: HA (aq) + OH (aq) A (aq) + HO (l). Notice that the Cl- was canceled out from the Net Ionic Equation, because it really isn't playing an important part of this reaction. This is called the Total Ionic Equation.ģ) Then, you simplify by canceling things out if they appear on both sides of the reaction, resulting in the Net Ionic Equation.Īnd other oxidation half and simply add themįor example, let look at the reaction of calcium carbonate with hydrochloric acid to form calcium chloride, water, and carbon dioxide:ĬaCO3(s) + 2HCl(aq) -> CaCl2(aq) + H2O(l) + CO2(g)ĬaCO3(s) + 2H+(aq) + 2Cl-(aq) -> Ca2+(aq) + 2Cl-(aq) + H2O(l) + CO2(g)ĬaCO3(s) + 2H+(aq) -> Ca2+(aq) + H2O(l) + CO2(g) 
Write the reaction out with all of the separated ions. If something is insoluble, it should not be broken apart. You will have to use the solubility rules to do this, they can be found online. This is also called the Molecular Equation.Ģ) Then, you break apart the soluble molecules into the two ions that are formed (one positive and one negative). The molecular equation shows each of the substances in the reaction as compounds with physical states written next to the chemical formulas. 
To write a Net Ionic Reaction, follow these 3 steps:ġ) Start by simply writing the overall balanced chemical reaction. Include states-of-matter under the given conditions in your answer. A Net Ionic Equation is a chemical equation for a reaction which lists only those species participating in the reaction. Write complete ionic and net ionic equations for each of the following reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
